Lewis Dot Structure For Seo2
SeO2 lewis construction is made up of selenium and oxygen cantlet. It is a 1-D polymer chain having alternate selenium and an oxygen atom. This article illustrates the properties associated with the SeO2 lewis structure.
SeO2 lewis structure involves 1 selenium cantlet and 2 oxygen atoms, both of which belong to group 16 of the periodic table. This means that both these atoms have 6 valence electrons. To complete their stability criteria both oxygen atoms will form a double covalent bond with selenium and selenium volition form an expanded octet.
Emphasizing SeO2 lewis structure then it is a colorless solid and the well-nigh ordinarily known compound of selenium. Information technology is white or creamy white in appearance and is a volatile crystal or crystalline powder with a pungent smell. Its structural appearance, unlike other members of grouping 16 of the periodic table, it is a 1-D polymer in the form of interlocked bondage of selenium (Se) and oxygen (O) atoms. Here each selenium atom is pyramidal in shape with the terminal oxygen atom. The bond length of the Se-O Bridge is more than than Se-O at the ends. The stereochemistry of the SeO2 lewis structure polymer chain is syndiotactic.
SeO2 lewis structure too shows the property of sublimation. The properties of SeO2 lewis structure alter on sublimation. Its structural shape changes from pyramidal to aptitude shape every bit observed in sulfur dioxide (Soii). SeO2 lewis structure is an acidic oxide. It can form selenous acid when dissolved in water. It can as well react with the base to grade neutral selenite salts. The chemical reaction is shown below:
SeOtwo + 2NaOH gives Na2SeO3 + H2O
SeO2 lewis structure presence in nature is quite rare. Its natural form is a very rare mineral found in the earth'south crust named downeyite. Information technology is unremarkably spotted in the vents formed during coal mines used for4 escaping gases. Other than that maximum product of SeO2 is washed artificially and synthetically in academic laboratories and chemical industries.
SeO2 preparation is very easy. The well-nigh common method of preparation is past oxidation of selenium (Se) by burning air:
Se + Oii gives SeOtwo
The other manner is past reaction with HNOthree (nitric acid) followed by dehydration of selenous acid.
3Se + 4HNO3 + H2O gives 3H2SeO3 + 4NO
H2SeO3 gives SeOii + HiiO
Hydrogen peroxide has even come in handy when preparing SeO2. The reaction of HiiO2 with Se gives SeO2.
2HtwoO2 + Se gives SeO2 + 2HiiO
SeO2 is a big game-changer in nanotechnology. When nowadays in the grade of actually tiny particles which cannot be seen with naked eyes, so it becomes a SeO2 nanoparticle which is a pre-requisite in many other scientific industries, especially the medical manufacture. These selenium dioxide nanoparticles are used as food additives. Many research papers and studies have shown that nano SeO2 acts every bit an antioxidant, and improves reproductive efficiency and fetal growth. It is also responsible for the growth of hair follicles. It has shown anti-bacterial and anti-viral properties whose evidence is mentioned in many scientific journals and bookish publications.
Apart from the medical industry, SeO2 is an important reagent in various organic synthetic methodologies. For instance, it is an important constituent of Riley Oxidation and is used to synthesize acylated hydrazone derivatives. SeO2 is besides used as a colorant in the glass industry. Information technology imparts a deep ruby color and is used as an active ingredient in cold bluing solutions. It also has usage as a toner in photographic development.
For understanding the structural representation of SeO2 lewis construction it is important to empathize the properties which contribute to its making. These properties are discussed in a detailed manner and are mentioned below:
- SeO2 lewis structure lone pair
- SeO2 lewis structure octet rule
- SeO2 lewis structure hybridization
- SeO2 lewis construction shape
- SeO2 lewis construction formal charge

SeO2 lewis structure lone pair
Lone pair of electrons dot not participate in bail formation past gaining, losing, and sharing. They are also called non-bonding electrons or unshared pairs of electrons. They are hands spotted in lewis structures as electron dots. The bonding ones are shown as lines or wedges.
In the SeO2 lewis structure, the lone pair central atom which is selenium is of importance because they make up one's mind the fate of many other properties. Lone pair of terminal oxygen atoms are ignored. So in the SeO2 lewis structure, the 2 electrons take shared either way with both the oxygen atoms forming a double covalent bond. So there is but a single lone pair of electrons on the selenium cantlet.

SeO2 lewis structure octet rule
The octet rule is a very important chemical dominion for the principal group elements. It emphasizes that each atom should have 8 valence electrons to achieve stability which is nearest to their noble gas configuration.
Talking virtually SeO2 lewis structure, then it comprises of single Se atom and ii oxygen atoms. Both these elements belong to group 16 of the periodic tabular array and have 6 valence electrons. Both the oxygen atoms can complete their octet stability by sharing their 2 electrons leading to double covalent bond formation.
Selenium on the other hand can adjust more than 8 electrons in its valence shell because of the presence of empty d-orbitals thereby forming an expanded octet. And so selenium in SeO2 lewis structure is an anomaly to octet rule and is a hypervalent species.
SeO2 lewis structure hybridization
Hybridization is the intermixing of orbitals to class new hybrid orbitals with similar energies merely different backdrop. As far as the SeO2 lewis structure is concerned the hybridization is sp3. The electronic configuration of Se in its ground state is [Ar]3d104s24p4. While forming a chemical bond during SeO2 lewis structure the Se atom volition exist in an excited land with electronic configuration [Ar]3d104s24p34di.
There will be the generation of 4 new hybrid orbitals due to the interest of one 2s orbital and iii 2p orbitals of both the oxygen atoms. Also, it is important to know that ane of iv orbitals is donated by adjacent SeO2 lewis structure as it does non exist in single molecular grade just in chained polymeric course.

SeO2 lewis structure shape
There is a common misconception that the SeO2 lewis construction is linear in shape due to its molecular formula representation. But the reality is very unlike and shape cannot be predicated solely based on the molecular formula. The presence of a double bond and lonely pair of electrons on selenium has a considerable effect on the shape of the SeO2 lewis construction. Information technology could be best explained past VSEPR theory.
According to it the presence of an equal number of solitary pairs on both the terminal oxygen atoms and the presence of symmetrical double bonds exert stiff and equal repulsive forces in both the directions. Due to this, the oxygen atoms on both the sides of the SeO2 lewis construction confront in a downward direction thereby providing a bent-shaped pyramidal geometry.
Some other evidence of the bent structure is that SeO2 is not a single molecule but a chain where there is a clear deviation in the bond length of bridged Se-O and terminal Se-O.

SeO2 lewis structure formal charge
To confirm the stability and electrical distribution of the molecule we tin can summate the formal charge on each participating cantlet. The formal charge can be calculated as:
Formal Charge = Valence electrons – Non-Bonding electrons – ½ X Bonding electrons
FC for O: 6 – 4 – four/2 = 0
FC for Se: 6 – 2 – 8/ii = 0
FC for O: 6 – 4 – iv/2 = 0
As the formal charge of all the 3 atoms involved is 0, this confirms the reliability and authenticity of the SeO2 lewis construction.
All the higher up-discussed properties are the perfect ingredients to brand the perfect and reliable SeO2 lewis structure recipe which is explained in detail:
How to draw SeO2 lewis structure
Selenium (Atomic number = 34 and electronic configuration = ii,viii,18,six) and oxygen (Atomic number = 8 and electronic configuration = 2,6) belong to the chalcogen family or grouping 16 of the periodic tabular array with 6 valence electrons.
To draw the lewis structure it is of import to summate the number of valence electrons which comes out to be 6 + 6×2 =24. The central atom in the SeO2 lewis structure is the one with less electronegativity. Here selenium is the least electronegative atom. So it will be the central atom with 2 concluding oxygen atoms.
To complete their octet stability both the oxygen atoms will share their ii electrons with the selenium atom forming a double covalent bail. This volition lead to octet completion in terminal oxygen atoms. Selenium on the other hand will have ten electrons in its valence shell. This is because selenium tin adjust more than eight electrons in its valence shell due to the availability of d orbitals. This is anomalous behavior and is called an expanded octet. The species which prove this behavior is called hypervalent species.
The reliability and actuality of structure can be calculated with the assist of the formal charge of each atom involved. If information technology comes out to exist 0 then it confirms the authenticity and correctness of the structure. Over here as mentioned in a higher place the formal charge of the SeO2 lewis structure is 0.
The detailed explanation of the SeO2 lewis construction can throw light on many other physical and structural properties. The well-nigh pregnant ones are:
- SeO2 lewis structure resonance
- SeO2 lewis structure polarity
SeO2 lewis construction resonance
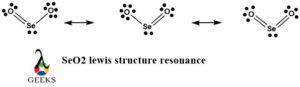
There is no fixed structure of the SeO2 lewis structure. Unlike many other lewis structures, it is a resonance hybrid of three structures. In these three structures of SeO2, there are 3 domains with i lone pair and 2 bond pairs that keep on aquiver. And then these shifting of electrons and changes in the location of bail pairs provide united states of america with 3 SeO2 resonance structures with near equal energies and stability. Here a single structure cannot explain all the desired properties of the SeO2 lewis structure. It can exist best explained diagrammatically.
SeO2 lewis structure polarity
Polarity is the behavior that is formed due to the separation of charge of 2 atoms which are together in chemic bail formation. In the context of SeO2 lewis structure and so it is a polar entity despite having a uniform formal charge distribution. Its dipole moment is 2.62D.
The polar nature of SeO2 is because of the difference in the electronegativity of Se and O which is greater than 0.5. The electronegativity of Se is 2.55 and oxygen is iii.44 which gives the divergence as 0.89. As it is more than than 0.5 so SeO2 lewis structure is polar.
SeO2 lewis structure (Related FAQs)
Explain the solubility of the SeO2 lewis construction.
Selenium dioxide is soluble in water. Forth with an aqueous medium, it shows solubility in organic solvents like benzene, acetone, and ethanol.
Depict the toxicity of SeO2.
SeO2 is not considered good for pulmonary health. It tin cause irritation, breathlessness, and cough. High exposure tin can atomic number 82 to the building up of fluid in the bronchial arrangement and can crusade pulmonary edema. Many other signs and symptoms are observed on accidental inhalation of SeO2 like nausea, sore throat, vomiting, and fatigue should not be ignored.
Also, please click to know virtually Characteristics of Metalloids.
Lewis Dot Structure For Seo2,
Source: https://lambdageeks.com/seo2-lewis-structure/
Posted by: madsenreflonch.blogspot.com


0 Response to "Lewis Dot Structure For Seo2"
Post a Comment